ATEX formation and explosion risk
Remember that an ATEX explosion is an instantaneous, violent combustion reaction. It involves two reagents: an explosive product and oxygen from the air. The first preventive measure against explosion is to avoid the risk of ATEX formation by extracting and filtering explosive substances.
When does the risk of ATEX formation begin?
United Kingdom, 2010. A forklift truck collides with a pallet in a warehouse selling hygiene products in aerosol cans. An ATEX is formed, a fire breaks out, then 80 seconds after a first explosion, then 150 seconds after a second explosion, destroying the entire warehouse.
The risk of forming an explosive atmosphere begins as soon as a combustible product is introduced onto the industrial site, or as soon as a combustible substance is produced during the transformation of a non-combustible product.
As a safety measure, the employer considers that any operation on site generates the probability of an explosive substance being released into the atmosphere. The explosive substance is released in the form of flammable gas, flammable liquid vapour or combustible dust. Each explosive substance has its own way of forming an ATEX.
How does the risk of ATEX formation progress?
The risk of forming an explosive atmosphere is confirmed by a succession of events increasing the concentration of the explosive substance in the air.
Let’s start from a situation where there are no combustible substances in the air. The kinetics of the risk include: the presence of the combustible product and the oxygen in the atmosphere, then the effective dispersion of the fuel in the atmosphere, followed by the beginning of homogenization. The kinetics depend on the characteristics of the combustible substance (density, granulometry…) and the characteristics of the atmosphere (turbulence, temperature, pressure…).
These events make the gas mixture potentially explosive. They change the concentration of the explosive substance in the atmosphere. But the combustion reaction will not occur as long as there is an unbalanced proportion between the two chemical reactants. Since the atmosphere cannot explode, we are dealing with an explosive atmosphere.
Explosive range: all non-explosive concentrations of a combustible product in the atmosphere. It characterizes the progression of the risk of ATEX formation.
When does the risk of ATEX formation arise?
The risk of ATEX formation arises when the concentration of fuel is such that the gas mixture becomes effectively explosive. At this point, the mixture is neither too lean nor too rich in fuel, and the proportions of fuel and oxidizer are sufficiently balanced to produce a combustion reaction. At this point, the fuel concentration is within the explosive range. The risk of explosion begins.
Explosive range : set of explosive concentrations of a combustible product in the atmosphere. Area where the gas mixture forms an ATEX and could explode under specific conditions.
The ATEX risk is characterized by the explosive range and the explosivity range.
The risk of ATEX formation is the precursor of the product-related risk of ATEX explosion. At the same time, it contributes to it. The explosive and explosive ranges characterize these risks.
Progression of combustible product concentrations from 0% to 100% in air
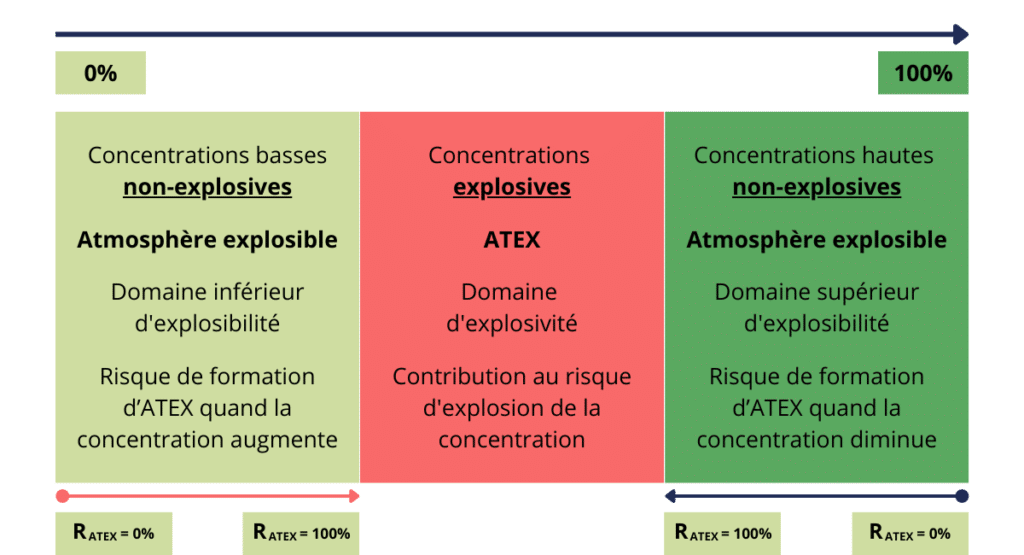
One way of preventing the risk of explosion is to keep the concentration of an explosive substance outside its explosive range. To reduce or eliminate the risk of ATEX formation, the employer must reduce or eliminate the concentration of the explosive substance in the lower explosive range. Suction and filtration of the explosive substance contribute to these two objectives.
Industrial processes sometimes require the use of high concentrations of explosive substances. Preventing the risk of ATEX formation means keeping their concentration in the upper explosive range, and as far away from the explosive range as possible.sivity. In this case, the use of an inert substance to replace oxygen is the most commonly used measure for preventing the risk of ATEX formation.
