As we have seen, the LEL and UEL define the explosive range, which consists of all explosive concentrations of an explosive substance in the atmosphere. To assess the risk, the employer will evaluate the concentration of the explosive substance based on the quantity used, and compare it to the explosivity limits. As a preventive measure, they will establish a safety margin, then dilute the concentration at least to the safety concentration using an extraction system/filtration of the ATEX.
Sommaire
- The LEL is used to relate the risk of ATEX occurring to the quantity of product in use.
- The explosive range, LEL and UEL provide information on the formation of ATEX, and the extent of the explosion risk associated with the product.
- The explosive range is used as a safety margin to prevent the formation of ATEX.
The LEL is used to relate the risk of ATEX occurring to the quantity of product in use.
The Lower Explosive Limit is compared to the concentrations of explosive substance that could form an ATEX. These concentrations depend on the emission rate of explosive substances. This itself results from the quantity of products present in the operational situation under study. The employer will estimate these concentrations for various ATEX formation scenarios.
A theoretical example: that of an ATEX gas leak scenario from a pipeline. The emission rate of the flammable product into the ambient atmosphere depends on the flow rate in the pipeline. This leads to the calculation of concentration gradients in the workspace. The concentration values are positioned in relation to the explosive range. From this, the degree of ATEX risk is deduced. The permanence of this risk is evaluated, according to the position in the workspace. The ATEX zones are then determined. Finally, preventive measures are applied, such as the extraction filtration of flammable gas. The extraction flow rate takes into account the potential leakage rate of the flammable gas.
A real-world example: in 2011, a 1000 m3 paper pulp tank, 95% full, exploded in Nogent sur Seine, causing one fatality. The paper pulp emitted hydrogen into the tank’s atmosphere. The emission rate per unit of dry mass is 20 dm3 per hour (pulp dryness 10%). At this rate, the LEL is reached after 1.6 hours. The hydrogen mixed with the
The explosive range, LEL and UEL provide information on the formation of ATEX, and the extent of the explosion risk associated with the product.
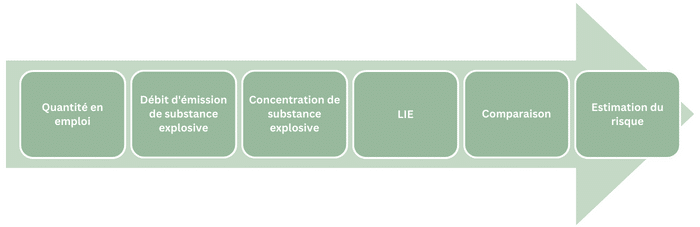
LEL and UEL specify the amplitude of the explosive range, the lower and upper explosibility ranges (ranges of non-explosive concentrations of an explosive substance). Consequently, they provide information on both the probability of ignition and explosion of the ATEX, and the probability of ATEX formation.
Thus, the wider the explosive range, the narrower the ranges of non-explosive concentrations become. Therefore, an emission of an explosive substance will have a higher probability of producing a concentration within the explosive range.
Furthermore, the wider the explosive concentration range, the higher the probability of explosion risk. (Note that the probability of explosion risk depends on other factors related to the atmosphere and the industrial environment.) When the recommended preventive measure is extraction filtration, the ATEX extraction flow rate partly depends on the concentration of explosive substance in that atmosphere. (Note that the concentration of explosive substance is not the only factor determining the extraction flow rate required to ensure the safety of the area.)
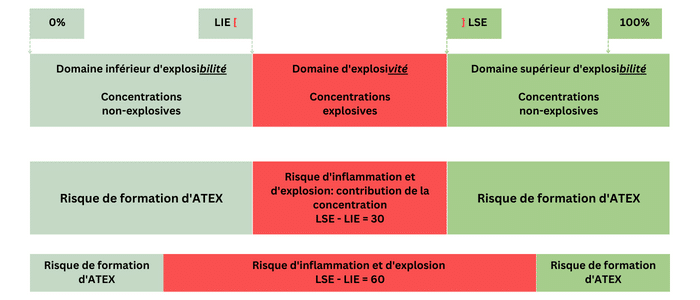
For example, the explosive range of hydrogen is 71 percentage points (4% to 75% by volume in air), while propane is 7.8 percentage points (2.2% to 10% by volume in air); approximately 10 times less. Therefore, there are more opportunities to encounter explosive situations with hydrogen.
And the time it takes to dilute the ATEX by suction to reduce the ATEX risk will be longer the closer the substance is to the upper explosive limit .
Furthermore, for the same amplitude of the explosive range, the lower the LEL (or the higher the UEL in the case of an oxygen-depleted atmosphere), the smaller the lower (or upper) explosibility range. Consequently, the faster the transition from a non-explosive concentration to an explosive concentration. Therefore, the lower the LEL, the higher the risk of ATEX formation.
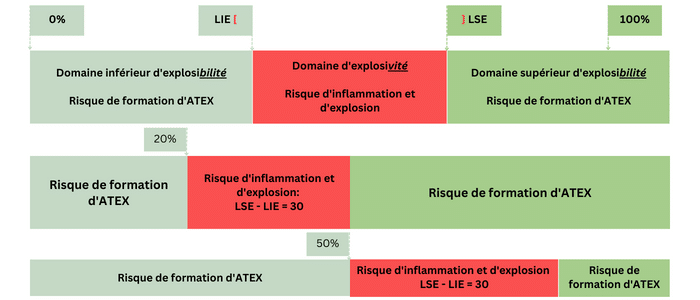
For example, if the initial situation is an atmosphere without combustible substance, then propane (LEL = 2.2%) presents a higher risk of ATEX formation than hydrogen (LEL = 4.4%). However, the risk of ignition and explosion is lower.
Under most operating conditions, it is easier to maintain the concentration below the LEL. To achieve this, extraction/filtration of combustible substances is the usual preventive measure. The lower the LEL, the higher the extraction flow rate must be, either to exit the explosive range, or to reduce explosive concentrations until the substance is eliminated from the atmosphere.
Some industrial processes rather require maintaining the explosive product at a concentration above the UEL. For example, cleaning processes involving immersion in a flammable solvent in a confined atmosphere.
The explosive range is used as a safety margin to prevent the formation of ATEX.
To prevent ATEX formation, it is necessary to keep the concentration of the gas or vapor outside the explosive range. Depending on ambient conditions, an explosive atmosphere will become more or less homogeneous in combustible products. For example, due to turbulence, certain areas considered outside ATEX could acquire an explosive concentration.
Thus, in ATEX risk prevention, the employer will take a safety margin on the explosivity threshold values. In this regard, ATEX regulations recommend establishing the concentration at a minimum of 10% below the LEL in the workplace where ATEX could form. And below the 25% threshold in other premises.[i]
[i] Circular of 09/05/85 on the technical commentary of decreesnos. 84-1093 and 84-1094 of 7/12/1984 concerning workplace ventilation and sanitation.
